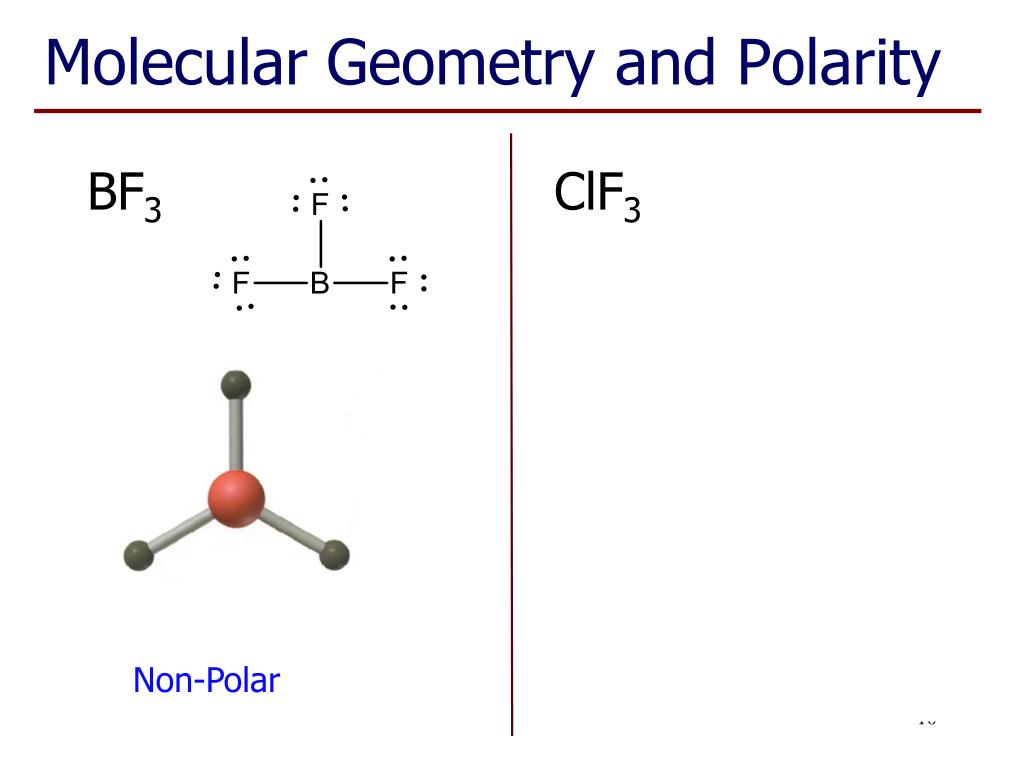
The dipole bonds are equal in magnitude but opposite in nature. It depends on the polarities of individual bonds and the geometry of the atoms. 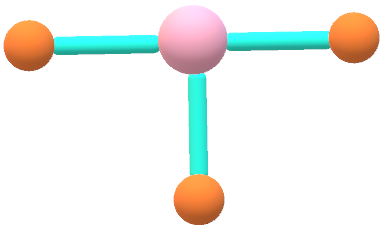
Zero dipole moment is the dipole moment between two atoms being zero. Dipole moment is measured in Debye units, which is equal to the distance between the charges multiplied by the charge. The size of the dipole is measured by its dipole moment$(\mu )$. When two electrical charges, of opposite sign and equal magnitude are separated by a distance, an electric dipole is established. Now, let’s try to define dipole moment significantly! Well, that was just an introduction to dipole moment! Let me make it a bit simple, Dipole moments occur when there is a separation of charge, this separation of charge can occur between two ionic bonds or between atoms in covalent bonds. This occurs when one atom is more electronegative than another, resulting in that atom pulling more tightly on the shared pair of electrons, or when one atom has a lone pair of electrons and the difference of electronegativity vector points in the same way. When atoms in a molecule share electrons unequally, they create what is called dipole moment. It is also a catalyst used in the reaction of alkylation, esterification and condensation reactions.įirst, let’s understand what dipole moment actually is! It’s an important Lewis acid very used in organic synthesis. This pungent colorless toxic gas forms white fumes in moist air.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
